Low Migration Inks and Coatings

INX International is a market leader in the development of low migration inks and coatings designed for food safe packaging applications. Labels and packages for foods and beverages and other printed items that have direct or indirect contact with food are the most important applications for Low migration inks. Using them reduces food packaging migration which can lead to food contamination. INX International offers a wide array of low migration inks and coatings for food safe packaging.
What is Food Packaging Migration?
Migration is the transfer of various components from the printed to the unprinted, product-facing side of the packaging substrate. These components can originate from inks, adhesives, varnishes, coatings, substrates, or the environment.
The term “low migration” packaging is commonly used to designate materials used in the whole packaging structure whose chemicals will not migrate from the packaging into the inside packed product.
Low migration inks and coatings are specifically designed for food safe packaging applications and have gone through migration testing to meet target compliance requirements. When using a low migration ink, it is the responsibility of the end user to determine the suitability of the final package construction by appropriate migration testing in accordance with the governing regulations or industry guidance. All possible sources of migration and contamination should be evaluated for compliance.
Here are five food-safe packaging tips.
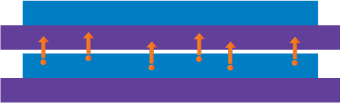
| How Ink Migration Can Occur From Printing Three common paths where migration can occur: |
|
|---|---|
| Physical Migration |
Penetration migration from printed side through substrate onto unprinted side
 |
|
Set-off migration from printed side to unprinted side of another sheet in a stack or roll
 |
|
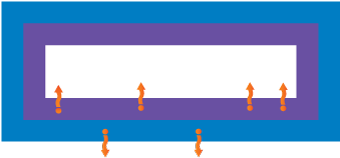
| Gas Phase Migration |
Evaporation or condensation migration, either due to the evaporation of volatile materials by heating (e.g. cooking, baking, or boiling frozen products in their original packaging) or the condensation through steam distillation
 |
LOW MIGRATION SOLUTIONS
INX International has worked for many years on understanding the factors affecting packaging migration and has developed a complete product portfolio of low migration inks and coatings to support the packaging chain to meet food packaging standards across a range of print processes and applications. We are committed to full compliance with the "EuPIA Guideline on Printing Inks applied to the non-food contact surface of Food Packaging & Articles”.
Our low migration inks and coatings include:
- Energy curable flexographic and offset inks
- High-performance sheetfed inks
- Energy curable coatings
- Oil-resistant gas barrier coatings
- Energy Curable Screen Inks
- Energy Curable Dry Offset Inks
INX Low Migration Brochure
The latest, most relevant regulations and standards, including FDA (U.S. Food and Drug Administration) requirements for the U.S., Canada, and Europe.
- Migration in packaging
- Supply chain roles
- Legal and regulatory Environment
- Practical recommendations
- FAQ’s
- Abbreviations, definitions, and resources
Not sure what product is right for your application?
Download the Low Migration Product Selector Guide